Featured Firms
Presented by BigVoodoo
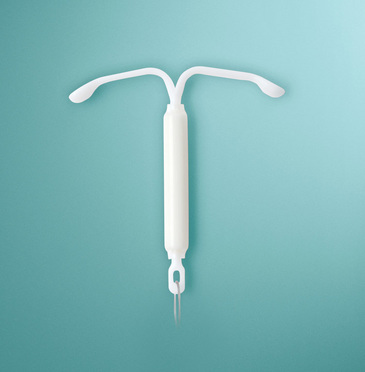
The U.S. Court of Appeals for the Second Circuit upheld Tuesday a summary judgment dismissing the multidistrict lawsuit against Bayer Pharmaceuticals, the manufacturer of the intrauterine birth control device Mirena.
October 24, 2017 at 04:09 PM
1 minute read
Presented by BigVoodoo
The New York Law Journal honors attorneys and judges who have made a remarkable difference in the legal profession in New York.
Law firms & in-house legal departments with a presence in the middle east celebrate outstanding achievement within the profession.
The premier educational and networking event for employee benefits brokers and agents.
Atlanta s John Marshall Law School is seeking to hire one or more full-time, visiting Legal WritingInstructors to teach Legal Research, Anal...
Shipman is seeking an associate to join our Labor & Employment practice in our Hartford, New Haven, or Stamford office. Candidates shou...
Evergreen Trading is a media investment firm headquartered in NYC. We help brands achieve their goals by leveraging their unwanted assets to...
MELICK & PORTER, LLP PROMOTES CONNECTICUT PARTNERS HOLLY ROGERS, STEVEN BANKS, and ALEXANDER AHRENS